All About Parabens
What are Parabens?
Parabens is a term used for para-hydroxybenzoates acid. Chemically, Parabens are esters. An ester is a compound formed from an alcohol and an organic acid (in the case of parabens, that acid is p-hydroxybenzoic acid). The most common parabens used in cosmetic products are methylparaben, propylparaben, and butylparaben. Parabens are the most widely used preservatives in cosmetics. Parabens are used to prevent the growth of yeasts, molds, and bacteria in cosmetics products, both to protect consumers and to maintain product integrity. Without them, bacteria and fungi would begin growing in these products, spoiling them and possibly harming individuals. Parabens are relatively easy to produce and incorporate into products, making them an ideal way to lengthen the shelf life of a variety of consumer goods.
What are Parabens?
Parabens is a term used for para-hydroxybenzoates acid. Chemically, Parabens are esters. An ester is a compound formed from an alcohol and an organic acid (in the case of parabens, that acid is p-hydroxybenzoic acid). The most common parabens used in cosmetic products are methylparaben, propylparaben, and butylparaben. Parabens are the most widely used preservatives in cosmetics. Parabens are used to prevent the growth of yeasts, molds, and bacteria in cosmetics products, both to protect consumers and to maintain product integrity. Without them, bacteria and fungi would begin growing in these products, spoiling them and possibly harming individuals. Parabens are relatively easy to produce and incorporate into products, making them an ideal way to lengthen the shelf life of a variety of consumer goods.

Parabens are chemical preservatives that have been identified as estrogenic and disruptive of normal hormone function. Estrogenic chemicals mimic the function of the naturally occurring hormone estrogen, and exposure to external estrogens could potentially lead to breast cancer. Parabens appear in some deodorants and antiperspirants. In addition, Parabens are found in personal care products such as shampoos, conditioners, lotions, and facial and shower cleansers and scrubs. Parabens are also found in foods such as salad dressing, processed vegetables, frozen dairy products, soft drinks and baked goods. They’re also widely used as preservatives in foods and pharmaceutical products. An estimated 75 to 90 per cent of cosmetics contain parabens. Nearly all of the parabens used as preservatives are man-made and not naturally occurring. However, some parabens are naturally created in foods such as blueberries, beans and cucumbers.
 Paraben where R= an alkyl group.
Paraben where R= an alkyl group.
Chemical Structure
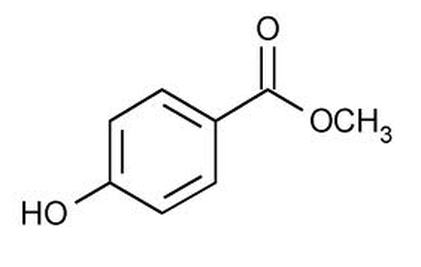
Methylparaben also known as parabens are used to make products last longer; their chemical properties make them effective preservatives that keep ingredients fresher for longer. The IUPAC name for the polymer of Methylparaben is Methyl 4-hydroxybenzoate. The chemical formula for methylparaben is CH3(C6H4(OH)COO). The Molecular Formula of methylparaben is C8H8O3 and the Molar Mass: 152.15 g/mol -1. The Functional groups located in the chemical are: Alcohol, Benzene, Alkane, and Ester. The ester group could be identified by the R Group: - O - C=O - (R Group).
Methylparaben also known as parabens are used to make products last longer; their chemical properties make them effective preservatives that keep ingredients fresher for longer. The IUPAC name for the polymer of Methylparaben is Methyl 4-hydroxybenzoate. The chemical formula for methylparaben is CH3(C6H4(OH)COO). The Molecular Formula of methylparaben is C8H8O3 and the Molar Mass: 152.15 g/mol -1. The Functional groups located in the chemical are: Alcohol, Benzene, Alkane, and Ester. The ester group could be identified by the R Group: - O - C=O - (R Group).

Chemical Reactions:
Parabens are a family of alkyl esters of para-hydroxybenzoic acid that fluctuate at the para arrangement of the benzene ring by different chemical substitutions of the side chains. The chemical substitutions provide each paraben ester with a different solubility. As the alkyl chain length increases, oil solubility increases and water solubility decreases. All parabens are unnaturally produced, although some are identical to those found in nature. They are produced through the esterification of para-hydroxybenzoic acid with the right alcohol, such as methanol, ethanol, or propanol. Para-Hydroxybenzoic acid is in turn produced scientifically from a modification of a reaction, using carbon dioxide.
Parabens are a family of alkyl esters of para-hydroxybenzoic acid that fluctuate at the para arrangement of the benzene ring by different chemical substitutions of the side chains. The chemical substitutions provide each paraben ester with a different solubility. As the alkyl chain length increases, oil solubility increases and water solubility decreases. All parabens are unnaturally produced, although some are identical to those found in nature. They are produced through the esterification of para-hydroxybenzoic acid with the right alcohol, such as methanol, ethanol, or propanol. Para-Hydroxybenzoic acid is in turn produced scientifically from a modification of a reaction, using carbon dioxide.